Absorption is used to remove one or more gaseous components from a gas flow using a solvent. Selective absorption is an important industrial process for the treatment of gas mixtures. CE 405 can be used to investigate the basic processes on the water-oxygen-nitrogen system.
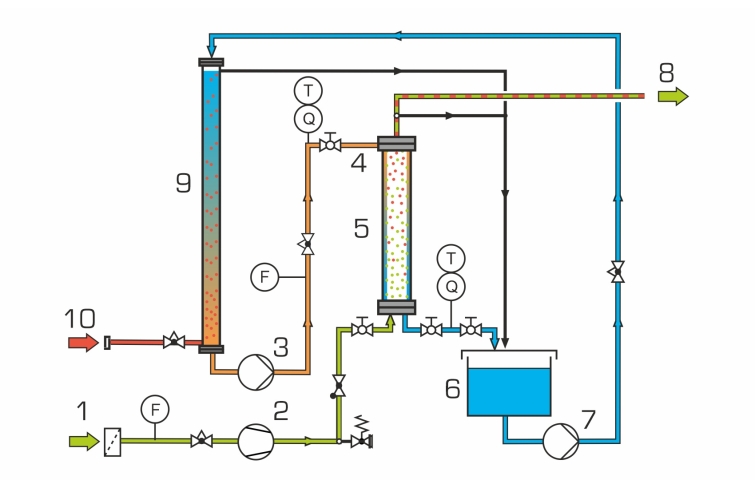
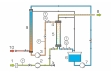
A compressor supplies ambient air from below into the absorption column. Water flows down as a thin film at the edge of the absorption column. The air flows upwards centrally in the column. A portion of the air’s oxygen is dissolved in the water film. The air flow exits the column at the top. The water containing the dissolved oxygen leaves the column at the bottom and flows into a tank. A pump supplies the water with the dissolved oxygen to the head of the desorption column.
The desorption column is a simple tube in which the water flows downwards. Nitrogen from a compressed gas cylinder enters at the base of the column. The nitrogen rises to the top in the form of dispersed bubbles in the water. The partial pressure of the oxygen in water is higher than the partial pressure in the gas phase (nitrogen). For this reason, a portion of the oxygen passes over from the water into the gas phase (stripping). This process leads to the water’s absorbing capacity for oxygen increasing. A pump supplies the solvent regenerated in this way to the head of the absorption column. Transparent materials allow optimal observation of the processes in both columns.
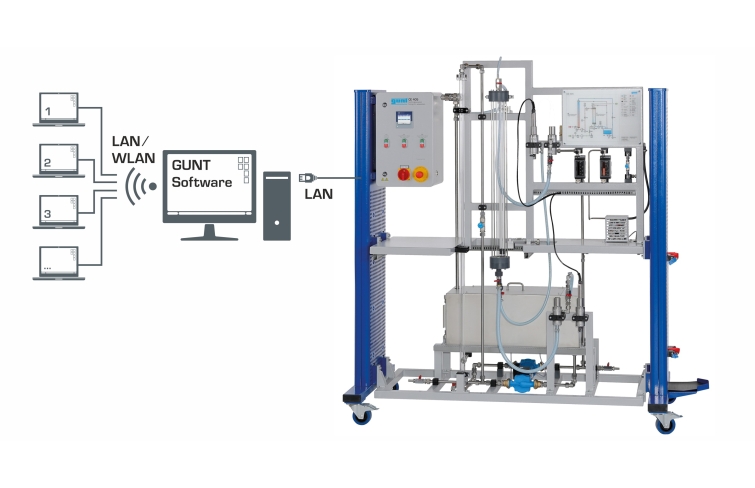
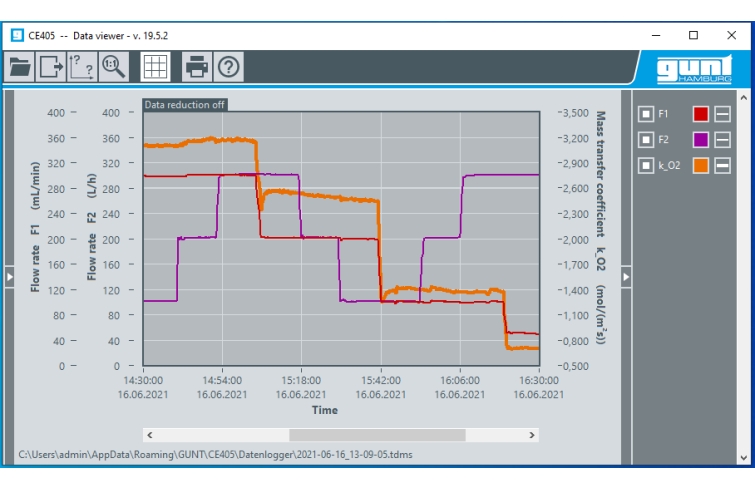
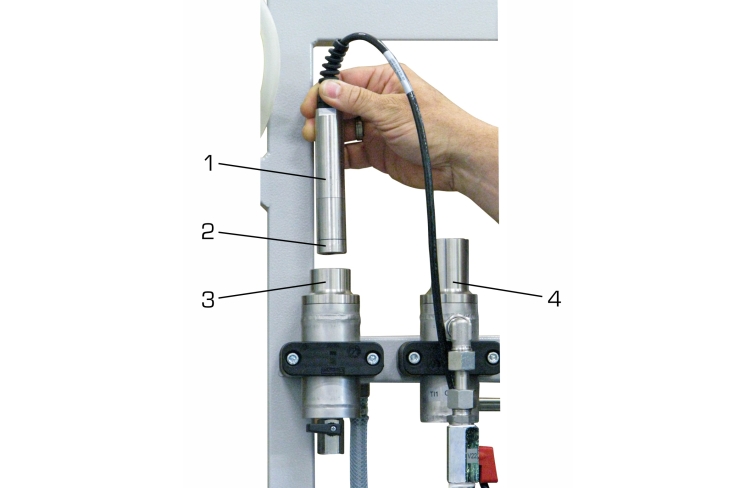
Valves and flow meters make it possible to adjust the flow rates of air and solvent. The oxygen concentration and temperature are continuously measured both upstream and downstream of the absorption column and digitally displayed. The measured values can be transmitted simultaneously via LAN directly to a PC where they can be analysed using the GUNT software.